Investigating the genetic basis of complex human traits is a pivotal endeavor in modern biomedical research. In a recent study, researchers at the Weizmann Institute of Science delved into a deep repository of clinical and genetic data to unravel the genetic associations across a range of disease phenotypes. Their innovative approach combines Genome-Wide Association Studies (GWAS) and Polygenic Risk Score-Phenome-Wide Association Studies (PRS-PheWAS) to analyze over 700 clinical phenotypes and 4,000 molecular features. From glycemic control to mental disorders, the study identified a number of significant genetic and phenotypic associations. Through trait filtering and genotype analysis, the researchers uncover novel genotype-phenotype associations, paving the way for a deeper understanding of complex conditions like obesity and diabetes.
Blog
Gencove Team - May 06, 2024
Genome-wide and phenome-wide associations: Advancing our understanding of the genetic contributions to health
This study is part of the “Human Phenotype Project” (HPP) which aims to combine multi-omic datasets for large populations to examine individual human genomes, microbiomes, transcriptomes, metabolomes, and more to unravel the complexity of human disease and identify novel diagnostic biomarkers and targets for actionable treatments or diagnostics.
What are quantitative phenotypes?
Quantitative traits are phenotypes that exhibit continuous variation within populations. They are often influenced by multiple genes and environmental factors, which result in a wide range of possible phenotypes; common examples include height, weight, blood pressure, and susceptibility to cardiovascular disease, among others. The liability to most diseases in humans with a genetic component is now understood to be highly polygenic, meaning that they are influenced by many genes, each of which has a small effect.
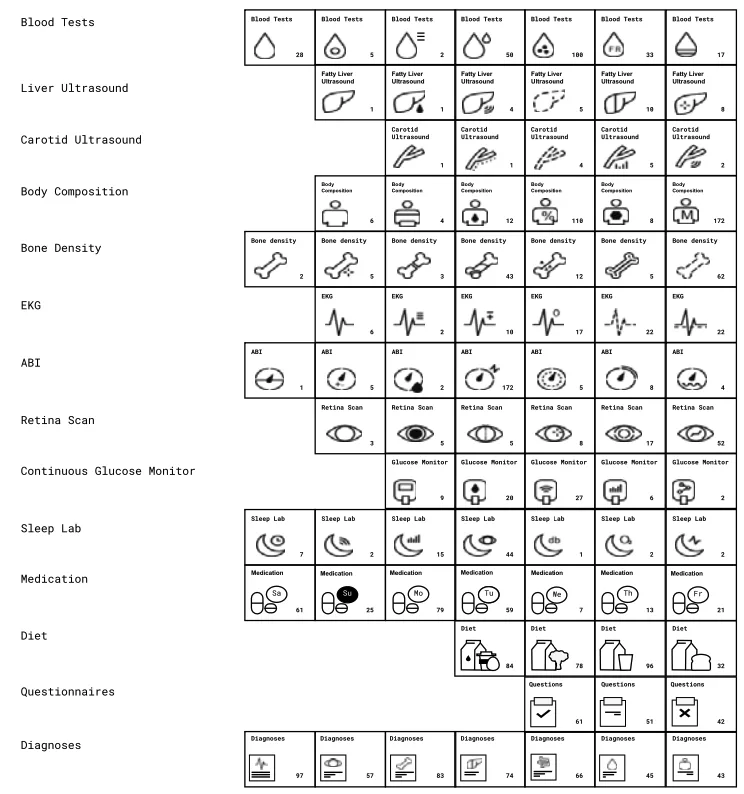
Statistical power is key for identifying potential biomarkers for complex traits, which is why the construction of such a vast database of over 8,680 individuals, containing genetic, clinical, and molecular data was necessary to reveal new associations for clinical and molecular features.
“We can use a whole spectrum of dynamic features that can yield insights and capture things that we may miss if we only look at individuals who have and don’t have disease” - Zachary Levine
Combining GWAS and PRS-PheWAS to uncover novel signatures
The study used low-pass whole genome sequencing to sequence DNA from buccal swabs of thousands of participants. Researchers then compiled the data for analysis using Gencove’s imputation pipelines. This genetic data was combined with clinical measurements, including results from imaging, questionnaires, and other health assessments, and subsequently analyzed to assess associations between genetic variation within the dataset and the clinical phenotypes.
The researchers turned to two trusted tools to determine the molecular and phenotypic relationships between the datasets – GWAS and PheWAS. GWAS is a common statistical method used to detect associations between genetic variation and phenotypes within a given cohort. Subsequent PRS-PheWAS analysis in which novel phenotypes collected in the study were tested against PRS for existing related phenotypes within the UK Biobank was performed to validate these novel phenotypes by showing that they associate with the genetic signal underlying existing measures.
The study revealed 169 clinical traits associated with 1184 SNPs, including genes associated with glycemic control as well as psychiatric disorders such as schizophrenia. Specifically, with metabolites and glycemic variance, the researchers found that there were 70 metabolites with at least one study-wide significant hit. The PRS-PheWAS portion of the study identified over 16,000 phenome-wide associations between previously derived PRS and the clinical phenotypes collected in this study including data for sleep patterns, liver health, vascular health, hormone regulation, mental disorders, and lifestyle factors answered by individuals who participated in the study.
You can access an interactive dashboard displaying the GWAS results here
Enhancing statistical power with imputation
The Weizmann Institute’s work with Gencove, and this resulting study demonstrates the power of combining low-pass sequencing and analysis pipelines with advanced data management and analysis tools. By mapping out the range of genetic determinants behind health and disease, this study paves the way for a deeper understanding of human biology, promising a future where genetic insights lead to more personalized, effective, and actionable healthcare solutions.