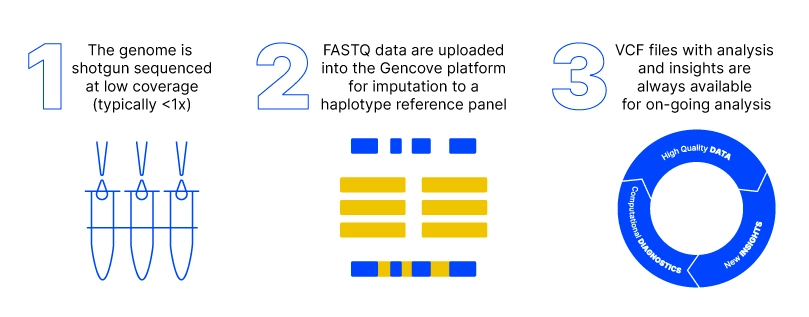
Pharmaceutical companies use Whole Exome Sequencing (WES) in their drug discovery and development process. However, R&D teams still need a comprehensive view of genomic variation outside the exome. Historically, the only way to augment WES were arrays, but now pharma companies have an alternative - low-pass whole genome sequencing.
Blog
Josh Setton, Head of Sales - Jun 09, 2022
Three Ways Gencove’s Low-Pass Whole Genome Sequencing is Modernizing the Drug Discovery Process
How low-pass sequencing helps pharma companies
From published data and real-world evidence, there are at least three points along the drug development pathway where low-pass whole genome sequencing is of particular benefit. We invite you to participate in this discussion and share the way you are, or would like to, apply cost-effective, high-throughput whole genome sequencing for therapeutic applications.
1. Discovery and development
Gencove’s low-pass sequencing helps to identify rare or novel variants by delivering high-quality, genome-wide data including high coverage at loci of interest. Because we can store your data, this enables flexible and ongoing downstream analysis.
Gencove’s platform is fast and affordable, alleviating time and budget pressures. In fact, our software is optimized from miniaturized library prep through advanced analysis for large-scale genomic projects.
Lastly, due to broad drug development candidate portfolios, low-pass sequencing allows pharma companies to standardize on one efficient and consistent way to augment WES and integrate all genomic data into current workflows.
2. Clinical trial design
Once the drug is ready for clinical trials, the medical and clinical operations teams appreciate using sequencing to ensure trial diversity and to determine the best genotype for patient selection.
3. Clinical research
Low-pass whole genome sequencing is an extremely efficient way to quantify safety and efficacy based on genomic test results. In fact, the Gencove platform accurately identifies genetic variants that influence drug response such as ADME genes. These data are particularly beneficial for the FDA applications to describe, predict, and analyze responder rates.
Are there additional ways low-pass sequencing can benefit the drug development process? Check out our Human genomics page to learn more.